
Research Topics
Lithium and Sodium Solid Electrolytes
As advanced energy-storage devices, lithium-ion batteries (LIBs) are becoming indispensable in its wide application of various electronic devices, electric vehicles, and renewable energy integration. However, conventional lithium-ion batteries (cLIBs) with organic liquid electrolytes have serious safety issues due to several risks of leakage and inherent flammability. In this regard, all solid-state batteries (ASSBs) using solid electrolytes are drawing attention as the next-generation energy storage. At the same time, lithium solid- state electrolytes provide a wider electrochemical window and good compatibility with high- potential electrodes, compared to liquid electrolytes, thus lead to higher energy density.


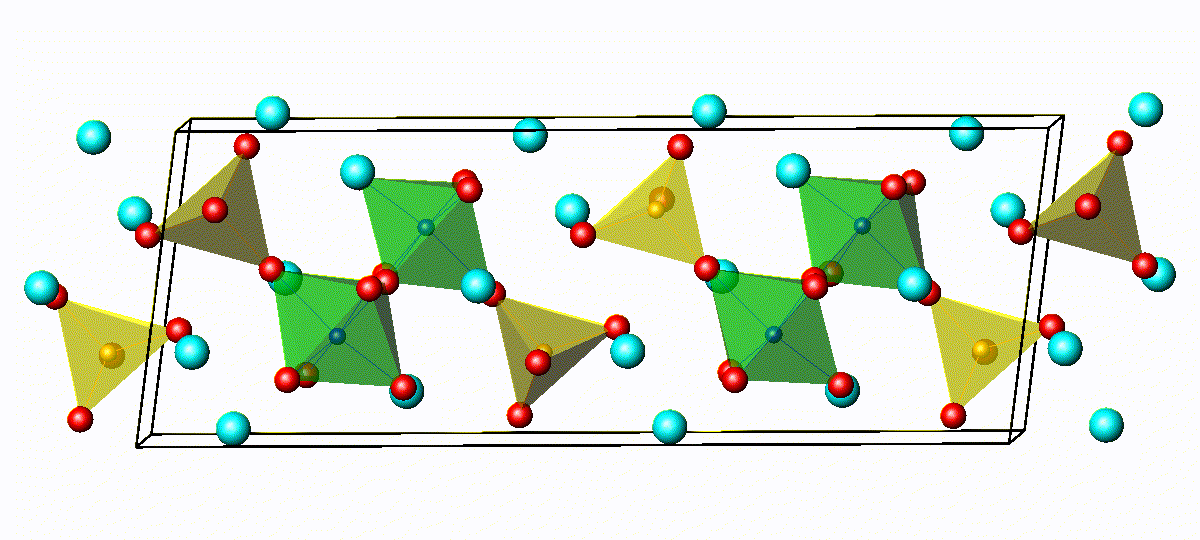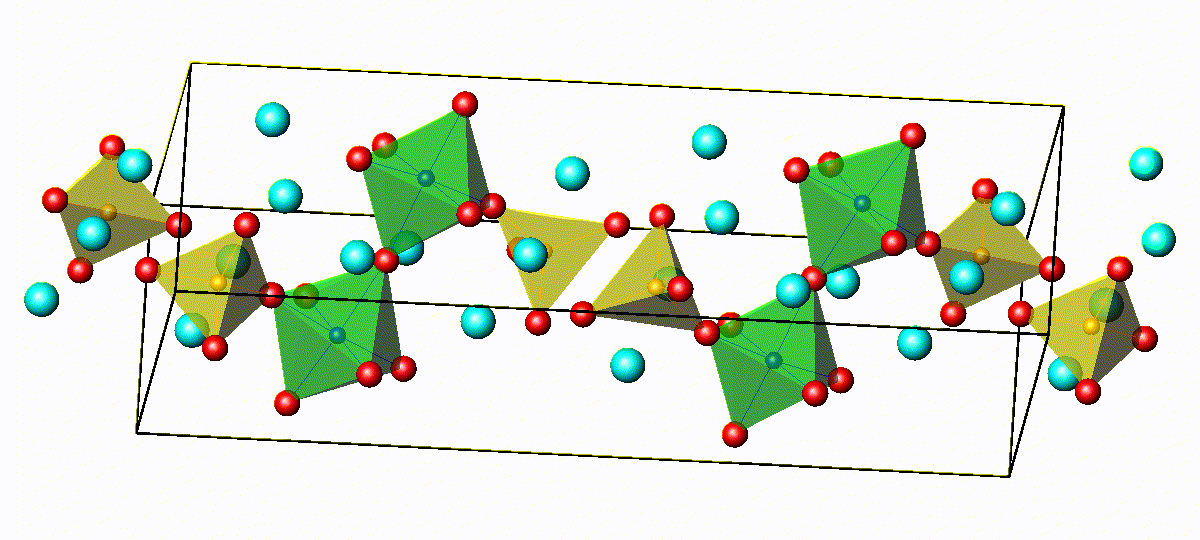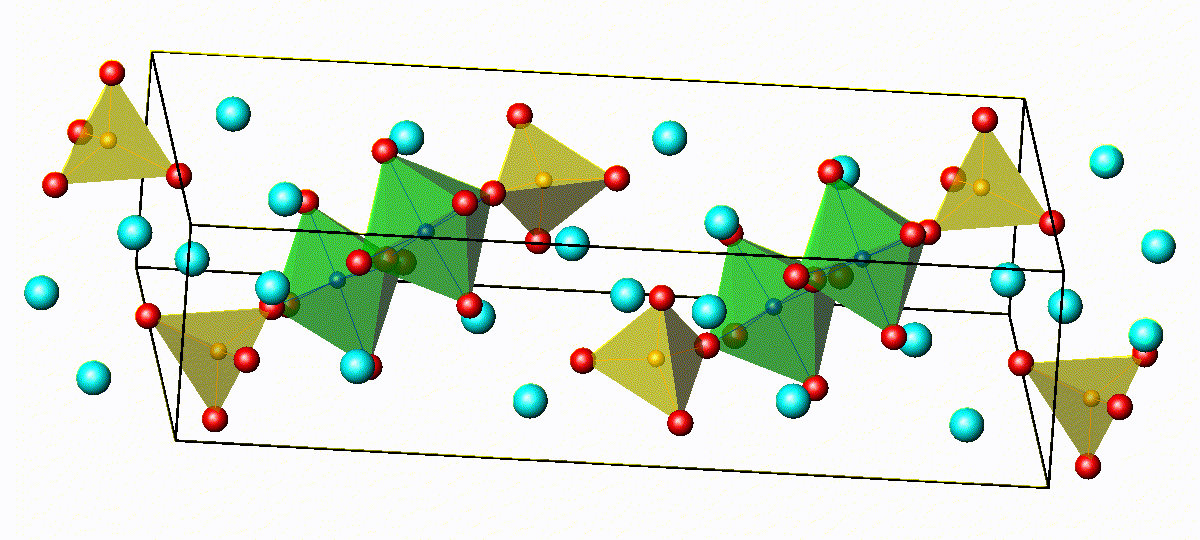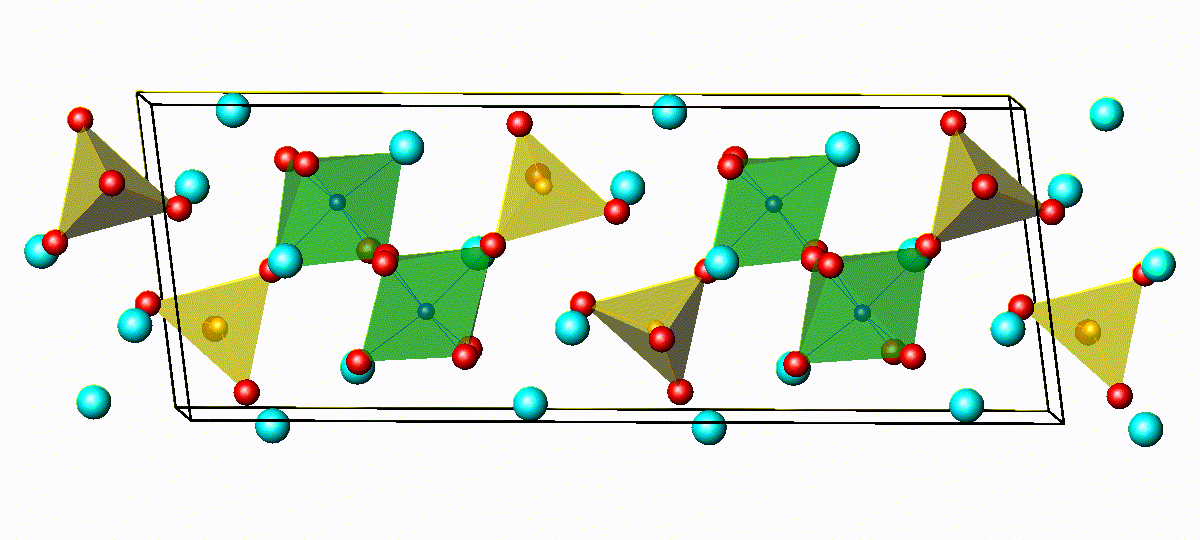
Crystal Structure of Na4−xSn1−xSbxS4
3D bond valence difference map isosurfaces
for Na4-xSn1-xSbxS4
Multivalent ion (Mg, Ca, Zn, Al) battery materials for post Li-ion batteries
Even though lithium ion batteries (LIBs) have been the most prevalent energy-storage technology so far especially in terms of energy and power densities, there are increasing concerns regarding the safety of Li-based batteries and the area preponderance of Li resources. The theoretical maximum energy density of LIBs based on the intercalation chemistry is expected not to be far exceed about 300 Wh/kg per cell. Since there are significant demands for higher energy densities and/or lower costs than those that LIBs can provide, multivalent ion batteries based on Mg, Ca, Zn and Al carrier ions have received attention due to its expected large volumetric energy density of their metals and the abundances on earth.


Abundance of elements on Earth's crust
(a) Illustrating ion and electron transports into a host structure for intercalation reaction, (b) The expected capacity increases when M(n+) ions intercalate into the same number of vacant sites of a host structure.
The objective of our researches is to discover and develop new battery chemistry and materials that could deliver higher energy densities and/or lower cost than those that LIBs can provide, through exploring new host materials and understanding electrochemical intercalation chemistry of the multivalent ions.
We search and design new electrode materials with a principle based on solid state chemistry, and discover completely new materials through exploratory synthesis.
The understanding of electrochemical intercalation chemistry (insertion mechanism, kinetics, energetics, structural changes etc.) during charge/discharge is the key to the development of the high-performing materials and batteries. One of our strengths is that we have advanced powder X-ray crystallographic techniques to study the details of the intercalation chemistry.

K and Na battery materials for ESS (energy storage system) aplications
With the development and popularization of smart grids and renewable energy generation such as solar and wind power, the rechargeable batteries have received much attention as energy storage systems. For such ESS applications, lower cost (per energy or power) and high durability are the most important factors to consider. Among several candidates, the sodium or potassium ion batteries are expected to provide cost-competitive systems.
The objective of our researches is to discover and develop new materials that could deliver lower cost than those that LIBs can provide, through exploring new host materials and understanding electrochemical intercalation chemistry of the sodium and potassium ions.
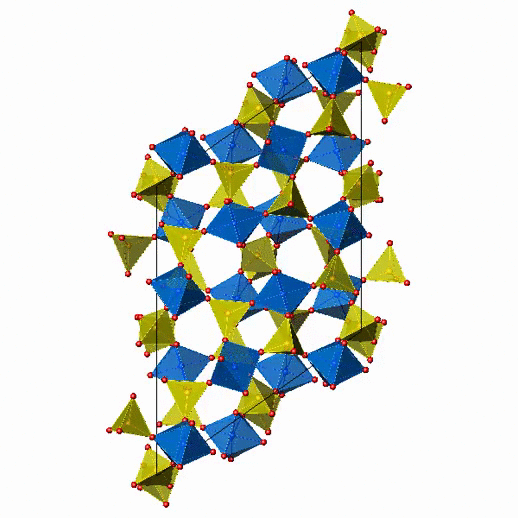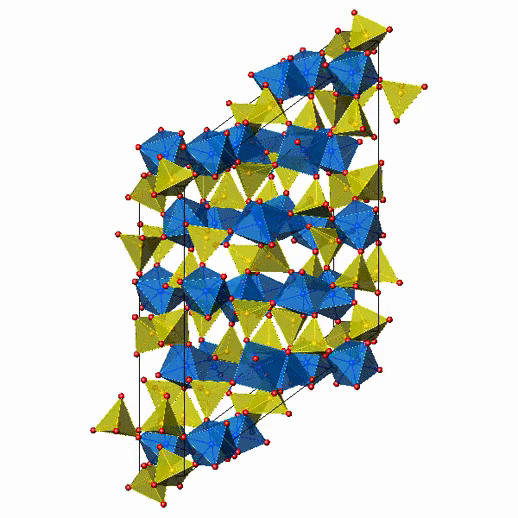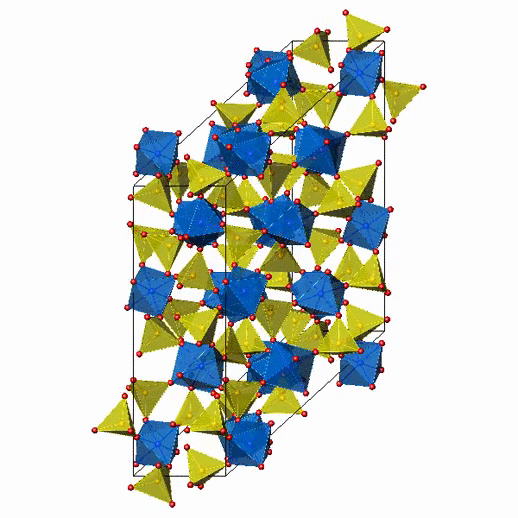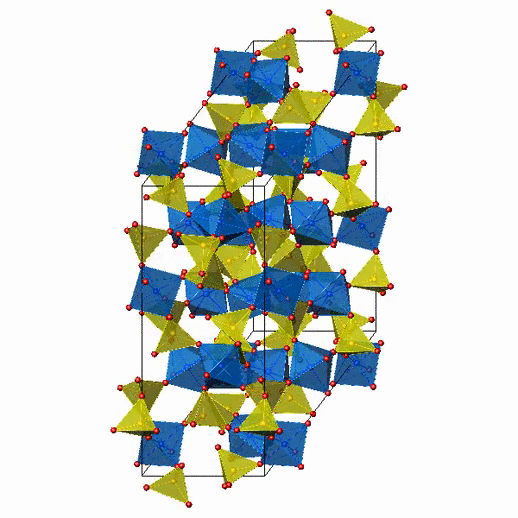

Crystal Structure of Fe2(MoO4)3
Cyclic voltammogram for Fe2(MoO4)3
with an electrolyte of 1 M Na(ClO4)2 in AN (0.05mV/s)

Determination of Na positions via electron density maps for the Na-inserted phase Na2Fe2(MoO4)3 from powder XRD data
High energy-density electrode materials for Li (metal) batteries
Even though the use of Li metal as an anode material is expected to increase the energy density of the batteries drastically, rechargeable Li metal batteries have been avoided for decades due to the safety concern related with the severe dendrite formation during cycling. Nevertheless, with the recent development of technologies to prevent or control the dendrite formation, it is likely that the rechargeable Li batteries will come back to the market in the next few years.
The cathode materials of present Li ion batteries MUST contain Li ions when they were prepared (e.g. LiCoO2, LiMn2O4 and LiFePO4) as the source of the carrier ion during cycling. For Li metal batteries, however, there is no such restriction on selecting the cathode materials. Thus, the choice of cathode materials is widely extended to compounds even without Li ions (e.g. MoS2, MoO3, V2O5).
The objective of our researches is to discover and develop new cathode materials for Li metal batteries that could deliver higher energy densities and/or lower cost than those that present LIBs can provide, through exploring new host materials intercalating Li ions.
Discovery of inorganic materials with new-to-the-world crystal structures
"It is always difficult to predict the unimaginable."
Prof. John D. Corbett
(Inorg. Chem. 2000, 39, 5178)
Discovery of new materials has been always the key to the advances in technologies or emergence of new applications. We are particularly interested in inorganic solid materials with unprecedented new-to-the-world crystal structures. It is, however, always difficult to predict or design the unprecedented or the unimaginable. That is why there have been relatively few discoveries of such kind. The objective of our researches is to discover such unprecedented materials using so called 'exploratory synthesis'.
The discovery of materials cannot be completed without structure determination. If a single crystal is available, the determination is straightforward using the classical crystallographic techniques. Unfortunately, a single crystal is hardly available for most of cases especially when you have a compound with a new structure in a mixture of several phases. In this case, the determination of crystal structure becomes extremely difficult. We have established our own method of structure determination from powder X-ray diffraction data, enabling us to discover new-to-the-world materials.
One of the research areas is phosphor materials research for White LED (WLED) applications, where such new-to-the-world crystal structures are highly important, but hard to get. We are applying our advanced synthetic and crystallographic techniques to exploring for new phosphor materials.
Crystal Structure of Sr4AlNbO8

Crystal Structure ofSr2Mg3P4O15

Crystal Structure of Mg(ClO4)2

Crystal Structure of Ba(ClO4)2